Bragg diffraction8/9/2023  
This data was collected at a fixed scattering angle. The experimental data above, reproduced above Davisson's article, shows repeated peaks of scattered electron intensity with increasing accelerating voltage. This peak indicated wave behavior for the electrons, and could be interpreted by the Bragg law to give values for the lattice spacing in the nickel crystal. It was a great surprise to them to find that at certain angles there was a peak in the intensity of the scattered electron beam. Their electron detector (called a Faraday box) was mounted on an arc so that it could be rotated to observe electrons at different angles. The electron beam was directed at the nickel target, which could be rotated to observe angular dependence of the scattered electrons. Electrons from a heated filament were accelerated by a voltage and allowed to strike the surface of nickel metal. The Bragg law for diffraction had been applied to x-ray diffraction, but this was the first application to particle waves.ĭavisson and Germer designed and built a vacuum apparatus for the purpose of measuring the energies of electrons scattered from a metal surface. Putting wave-particle duality on a firm experimental footing, it represented a major step forward in the development of quantum mechanics. The Davisson-Germer experiment demonstrated the wave nature of the electron, confirming the earlier hypothesis of deBroglie. J., "Are Electrons Waves?," Franklin Institute Journal 205, 597 (1928) If incident angle is zero then you can apply Bragg diffraction condition in this case too, only you have multiple d.Davisson-Germer Experiment Davisson-Germer Experimentĭavisson, C. Now same conclusion can be drawn as above but here you will get a spectrum in single shot.Īgain if you change the crystal type from single to poly-crystalline then the spots will turn into lines but the conclusion do not change. The pattern of these diffraction spots gives you the crystal structure and spacing between these spots gives you the spacing of atomic planes. If you change the geometry from Bragg to Laue keeping everything else is same then you will get the diffraction spots corresponding to the planes meeting the diffraction conditions. and if that is what the question is asking the angle difference between two peaks is larger for shorter spacing of crystal planes. Now you will get two diffraction peaks for two orders. In this case if you change the incident angle and at some angle when wavelength matches you will get diffraction pattern. I have also assumed that the x rays are monochromatic. Where n is the order of diffraction, $\lambda$ is the wavelength of the x ray and d is the crystal spacing. As you know that Bragg diffraction condition yields 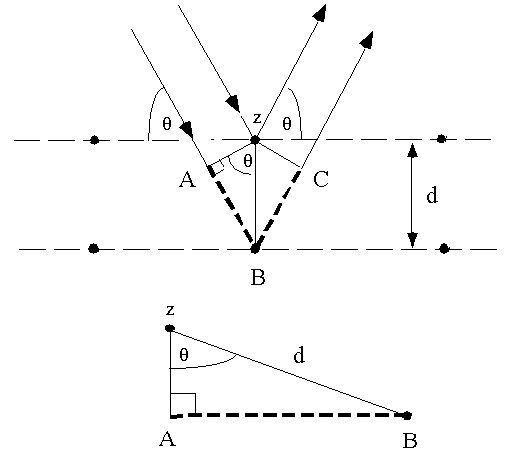
Since it is not mentioned anywhere I assume that it is a single crystal i.e. I suppose that crystal is in that mode only. In Bragg geometry the crystal is in reflective mode. Any input is appreciated :)Īs you know that there are two geometries of x ray diffraction namely Bragg geometry and Laue geometry. I have no idea whether my answer is correct, and I am also pretty certain I am overlooking a simpler (and more correct, given the phrasing of the question) way of solving the problem. I solved the problem graphically by drawing out a typical two wave source interference pattern (like two bobs dabbing on water in phase) and saw that smaller spacing between wave sources (atoms) $\iff$ larger spacing between maxima (of the same order) in the interference pattern. Are they referring to spots of maximum constructive interference that appear on a screen as a result of laue diffraction? If so, I am unaware of a way to relate the spacing between maxima to the spacing between atom layers (d). I'm not even sure what they mean by the question. I have been looking at this problem for quite some time now. "Based on the Bragg equation, if crystal A has larger spacing in its diffraction pattern than crystal B, what conclusion can you draw about the spacing between layers of atoms in A compared with B?" 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
